Uveitis Treatment May Aid Diabetic Patients
Review of Optometry, Vol. No: 143:4Issue: 4/15/2006
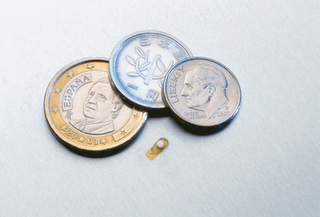
An intravitreal implant used to treat posterior uveitis also shows promise as a treatment for diabetic macular edema.
Retisert (fluocinolone acetonide intravitreal implant), releases a constant amount of fluocinolone, a corticosteroid, into the eye.
Preliminary follow-up data found that at 36 months after implantation, 58% of the implanted eyes showed no evidence of edema vs. 30% of eyes that received standard-of-care treatment. Also, 28% of the implant patients demonstrated a three or more line improvement in visual acuity vs. 15% of patients who received the standard of care.
The Retisert implant, shown here for size, is FDA approved for uveitis but also shows effectiveness in treating diabetic macular edema.
The most common adverse events included cataract requiring extraction in 95% of phakic implanted eyes and intraocular pressure rise in 35% of implanted eyes. Also, 36 eyes (28%) required a filtering procedure, and the device was explanted in six eyes (5%) to manage IOP.

An intravitreal implant used to treat posterior uveitis also shows promise as a treatment for diabetic macular edema.
Retisert (fluocinolone acetonide intravitreal implant), releases a constant amount of fluocinolone, a corticosteroid, into the eye.
Preliminary follow-up data found that at 36 months after implantation, 58% of the implanted eyes showed no evidence of edema vs. 30% of eyes that received standard-of-care treatment. Also, 28% of the implant patients demonstrated a three or more line improvement in visual acuity vs. 15% of patients who received the standard of care.
The Retisert implant, shown here for size, is FDA approved for uveitis but also shows effectiveness in treating diabetic macular edema.
The most common adverse events included cataract requiring extraction in 95% of phakic implanted eyes and intraocular pressure rise in 35% of implanted eyes. Also, 36 eyes (28%) required a filtering procedure, and the device was explanted in six eyes (5%) to manage IOP.



Comments
Post a Comment
Leave a Comment or Question: